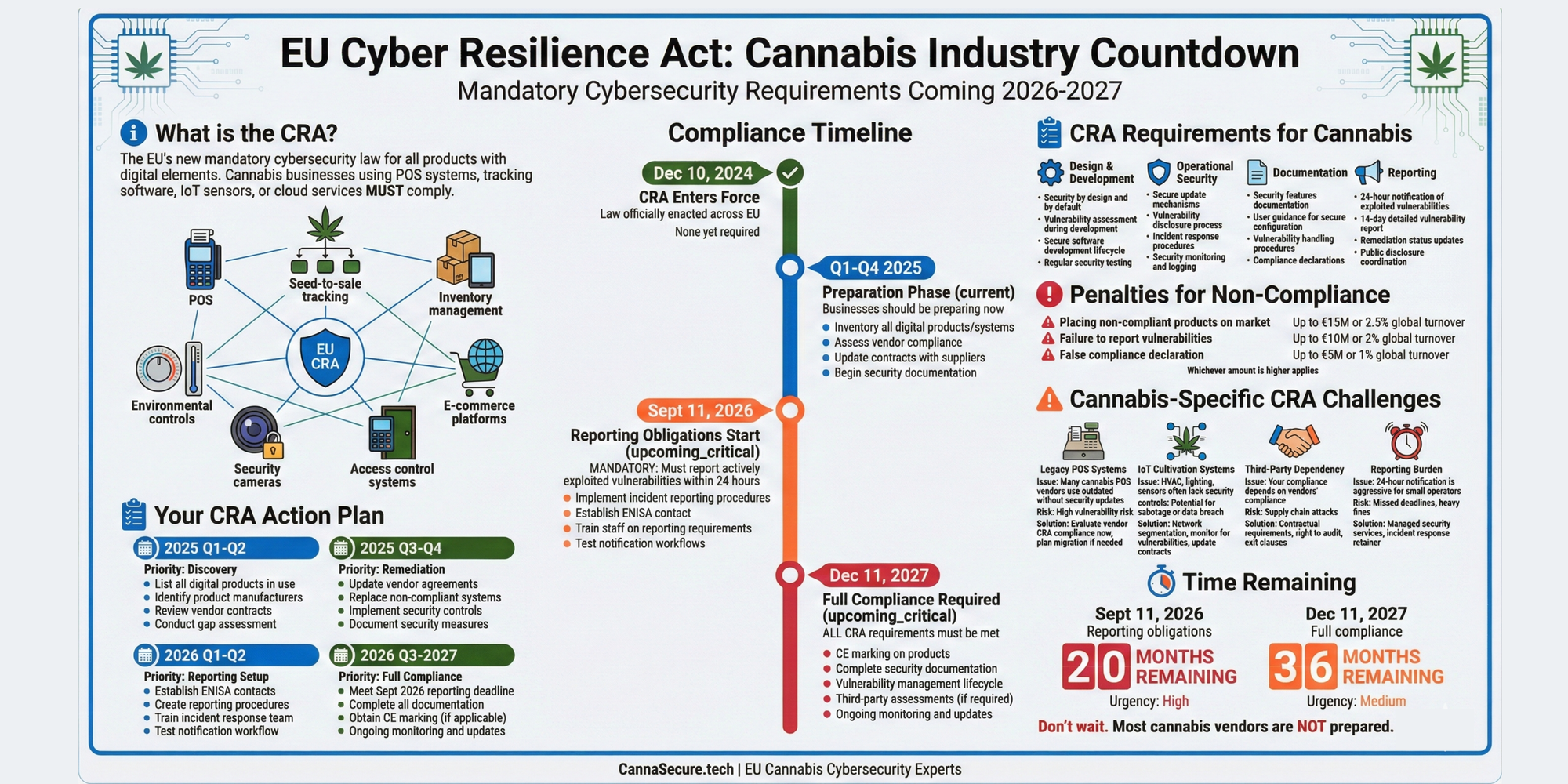
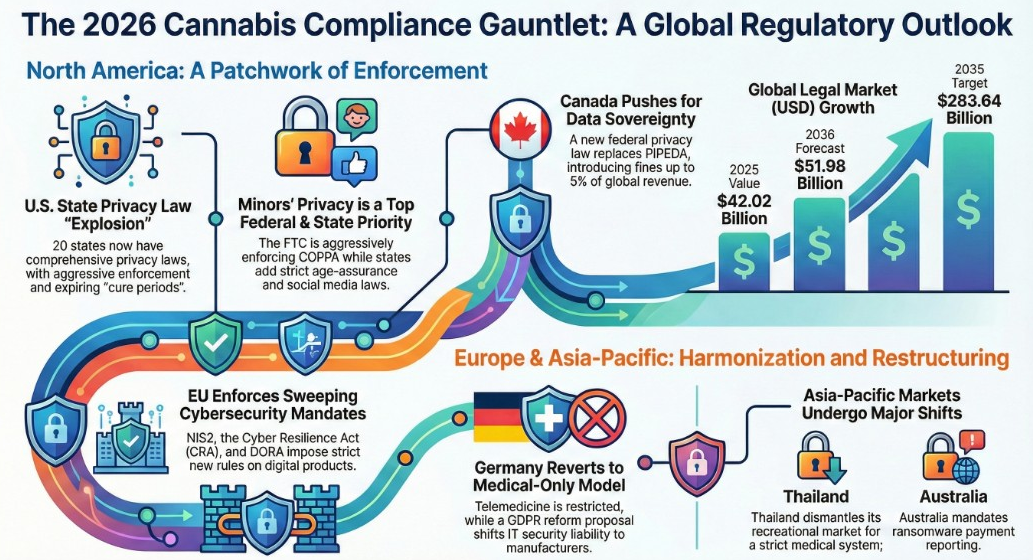
As France enters 2026, the nation’s cannabis regulatory landscape is undergoing its most significant transformation in decades. With the medical cannabis pilot program set to transition into a permanent pharmaceutical framework on April 1, 2026, and increasingly aggressive enforcement against synthetic cannabinoids, businesses operating in or exporting to France face a complex compliance maze that demands robust security protocols and real-time regulatory intelligence.
This comprehensive guide examines France’s evolving cannabis regulations through the lens of cybersecurity, supply chain protection, and regulatory compliance—critical considerations for businesses navigating one of Europe’s most restrictive yet potentially lucrative markets.
Executive Summary: Current Legal Status
France maintains some of the strictest cannabis laws in Europe, yet paradoxically remains the continent’s largest consumer market. As of January 2026, the regulatory environment is defined by several parallel frameworks:
Recreational Cannabis: Completely prohibited with €200 fixed fines (reducible to €150 if paid within 15 days), escalating to €3,750 fines and up to one year imprisonment for repeat offenses or larger quantities.
Medical Cannabis: Transitioning from 3,209-patient pilot program (ending March 31, 2026) to permanent pharmaceutical framework launching April 1, 2026. Currently limited to existing pilot participants with five approved conditions: treatment-resistant epilepsy, neuropathic pain, cancer symptoms, painful spasticity from MS/CNS disorders, and palliative care.
CBD Products: Legal with ≤0.3% total THC (increased from 0.2% in 2025). However, ingestible CBD products face Novel Food regulation challenges with zero fully authorized applications as of September 2025.
Synthetic Cannabinoids: Sweeping bans implemented throughout 2024-2025 covering HHC, HHCPO, H4CBD, THCP, THCA, H2CBD, and all substances with benzo[c]chromene chemical nucleus. Additional compounds including THV+ and THV-N10 under active regulatory scrutiny.
Critical Timeline: The March 31, 2026 pilot program end date represents a major inflection point, with full pharmaceutical framework implementation expected by Q2 2026 pending final EU approval and ministerial sign-off.
Recreational Cannabis: Penalties and Enforcement
Current Enforcement Landscape
France’s recreational cannabis prohibition operates through a multi-tiered penalty structure designed to reduce judicial burden while maintaining deterrent effect:
First-Time Possession (Small Amounts):
- €200 standard fine
- €150 if paid within 15 days
- €450 if unpaid after 45 days
- No criminal record for simple possession under new administrative fine system
Repeat Offenses/Larger Quantities:
- €3,750 maximum fine
- Up to 1 year imprisonment
- Criminal record
- Potential for suspended sentences in practice
Trafficking/Distribution:
- 5 years imprisonment
- €75,000 fine
- Article L3421-4 prohibits any “favorable presentation” of cannabis in media, advertising, or public discourse
Security Implications for Businesses
The prohibition framework creates several compliance and security challenges:
- Public Relations Risk: Article L3421-4’s broad interpretation criminalizes content that “presents narcotics in a favorable manner,” exposing media companies, publishers, and content creators to legal liability even for educational or news coverage.
- Cross-Border Transport: French customs enforce zero-tolerance policies at all entry points. Even CBD products from neighboring countries require extensive documentation (accredited COA, French labeling, batch tracing).
- Digital Surveillance: French law enforcement maintains sophisticated digital monitoring capabilities for cannabis-related commerce, requiring businesses to implement robust geo-blocking and age verification systems.
CBD Products: The 0.3% THC Compliance Minefield
Updated THC Threshold (2025)
France harmonized with EU regulations in 2025, raising the permitted THC limit from 0.2% to 0.3% total THC. However, this change introduced new compliance burdens rather than simplifying the regulatory environment.
Critical Compliance Requirements
Laboratory Testing:
- Accredited lab Certificates of Analysis (COA) mandatory for all imports
- Total THC measurement (not just delta-9 THC)
- French-language test results
- Batch-specific testing with unique identifiers
- Ongoing batch testing for domestic distributors
Packaging and Labeling:
- French consumer safety law compliance
- Complete ingredient listings
- Batch reference numbers
- Legal complaint address
- Warning labels
- No health claims (direct or implied)
Import Documentation:
- Original COA from accredited laboratory
- French translation of all documentation
- Proof of authorized hemp variety (Cannabis sativa L.)
- Supply chain documentation
- Customs declaration with HS codes
The Novel Food Enforcement Crisis
Critical Alert: As of September 2025, zero CBD Novel Food applications have received full EU authorization. This creates an existential threat for ingestible CBD products in France.
The DGCCRF (Direction générale de la concurrence, de la consommation et de la répression des fraudes) actively seizes, restricts, and recalls CBD products lacking validated Novel Food dossiers. Enforcement priorities include:
- Oils and tinctures
- Edibles (gummies, chocolates, beverages)
- Dietary supplements
- Any product intended for oral consumption
Compliance Strategy: Focus on non-ingestible formats (topicals, cosmetics, e-liquids, vaporization products) until Novel Food regulatory pathway clarifies. Monitor RASFF (Rapid Alert System for Food and Feed) for real-time enforcement actions.
Border Security and Seizure Risk
Even marginally non-compliant products face immediate customs seizure:
- Missing French labeling: Automatic seizure
- Incomplete COA: Seizure and potential criminal investigation
- THC overage (even 0.31%): Seizure with financial penalties
- Unapproved health claims: Market removal and sanctions
Security Recommendation: Implement pre-shipment compliance auditing with French regulatory specialists. The cost of rejected shipments far exceeds preventative compliance investment.
Medical Cannabis: From Pilot to Pharmaceutical Framework
The April 2026 Transformation
France’s medical cannabis program represents a unique pharmaceutical-centric approach, fundamentally different from medical marijuana frameworks in Germany, Netherlands, or North American markets.
Key Framework Elements (Effective April 1, 2026):
- Pharmaceutical Establishment Requirement: Only companies authorized as “pharmaceutical establishments” under French Public Health Code can apply for cannabis cultivation/distribution licenses. This eliminates traditional “cannabis industry” companies in favor of pharmaceutical operators.
- Temporary Use Authorizations (ATUs): ANSM (Agence Nationale de Sécurité du Médicament) oversees 5-year product authorizations with 9-month renewal windows before expiration. Initial approval period: 210 days.
- Indoor Cultivation Mandate: All domestic cultivation must occur indoors or in non-publicly-visible greenhouses with strict security protocols. Outdoor cultivation prohibited.
- Physician Certification: Only specially trained and certified physicians can prescribe. As of early 2025, only 274 specialists completed certification. Training programs expanding post-launch with HAS (Haute Autorité de Santé) oversight.
- Last-Resort Therapy: Cannabis remains treatment of last resort. Physicians must document failure or intolerance of all standard therapies before prescription.
- Sealed Vaporization Only: Raw flower permitted exclusively in sealed capsules for medical vaporization devices (Mighty Medic® or equivalent). Combustion prohibited.
Approved Medical Conditions
The permanent framework maintains pilot program’s narrow indication list:
- Treatment-resistant epilepsy
- Cancer-related symptoms (nausea, pain, appetite loss)
- Refractory neuropathic pain
- Painful spasticity from MS and CNS disorders
- Advanced palliative care (persistent unrelieved symptoms)
The Reimbursement Wild Card
Critical Uncertainty: Market viability depends entirely on reimbursement decisions expected by Q1 2026.
- SMR (Service Médical Rendu) Assessment: If HAS assigns “insufficient medical value,” products may be excluded from reimbursement entirely.
- CEPS Price Ceilings: Comité Économique des Produits de Santé may impose tight price controls limiting producer/distributor margins.
- Pilot Success Model: The pilot program succeeded because medicine was fully state-covered. Without comparable reimbursement, patient access collapses.
Market Projections:
- 2026: 5,454 patients, €9.6 million market (with reimbursement)
- 2035: 457,129 patients, €806 million market (Prohibition Partners estimate)
- However: These projections assume favorable reimbursement framework
Security Considerations for Medical Cannabis
The pharmaceutical framework demands enterprise-grade security infrastructure:
- Patient Data Protection: 3,000+ existing pilot patients transitioning to permanent registry. GDPR compliance mandatory with additional French health data regulations.
- Supply Chain Tracking: Seed-to-sale traceability requirements matching pharmaceutical standards. Blockchain or equivalent immutable ledger systems recommended.
- Facility Security: Indoor cultivation facilities require physical security meeting pharmaceutical manufacturing standards (access control, surveillance, environmental monitoring).
- Prescriber Authentication: Digital prescription systems must prevent fraud while maintaining patient privacy. Multi-factor authentication essential.
- API Security: Integration with ANSM authorization systems, pharmacy dispensing networks, and health insurance reimbursement platforms requires robust API security with encrypted data transmission.
Banned Substances: The Neo-Cannabinoid Crackdown
ANSM’s Expanding Ban List
France’s National Agency for the Safety of Medicines has implemented sweeping prohibitions on synthetic and semi-synthetic cannabinoids since June 2023:
June 2023 Initial Ban:
- HHC (hexahydrocannabinol)
- HHCO (HHC-acetate)
- HHCP (hexahydrocannabiphorol)
June 2024 Expanded Classification:
- All substances with benzo[c]chromene chemical nucleus
- H4CBD, H2CBD
- HHCPO (HHCP acetate)
- THCP (tetrahydrocannabiphorol)
- THCA (tetrahydrocannabinolic acid)
- THCB, THCJD, THCH
- THCV above 0.3% concentration
Under Active Scrutiny (Mid-2025):
- THV+ and THV-N10 (potential summer 2026 ban)
- 10-OH-HHC and 8-OH-HHC (HHC metabolites)
- CBN reclassification under consideration
The Rationale Behind Bans
ANSM cited multiple public health concerns justifying neo-cannabinoid prohibitions:
Psychoactive Risk: Benzo[c]chromene nucleus compounds demonstrate stronger CB1 receptor affinity than THC, producing:
- Intense euphoric effects
- Extended duration of action
- Unpredictable potency variation
- Higher addiction potential
Documented Adverse Events:
- Tremors, vomiting, anxiety
- “Bad trips” requiring emergency intervention
- Mental confusion, malaise
- Tachycardia, chest pain, blood pressure spikes
- Paranoia, panic attacks
- Cases of kidney failure (HHCPO)
- Convulsions, loss of consciousness, coma
Regulatory Evasion: Flexible molecular structures allow continuous modification to circumvent bans, creating enforcement challenges and detection difficulties.
Youth Marketing Concerns: Colorful packaging, sweet flavors, and candy formats targeting underage consumers.
Enforcement Priorities
DGCCRF and ANSM focus enforcement on:
- Products exploiting regulatory gray zones (rapid formula changes, “research chemicals”)
- Inaccurately labeled potency (especially misdeclared THC content)
- Youth-oriented products (gummy formats, cartoon packaging, dessert flavors)
- Online distributors (mystery shopping operations, influencer partnerships)
- Cross-border e-commerce (shipments from Netherlands, Spain, Poland)
Compliance Requirements for Businesses
Immediate Actions Required:
Comprehensive Audit: Remove ALL banned cannabinoids from inventory, product lines, and marketing materials. This includes:
-
Pure isolates
-
Blended products
-
“Proprietary blends” potentially containing banned substances
-
Products marketed under brand names (THM, THCN, MCPN, MCPB)
-
Enhanced Testing Protocols: Implement third-party testing for full cannabinoid profiles on all products. Standard CBD testing insufficient—must screen for banned synthetic cannabinoids.
-
Supplier Verification: Require sworn declarations from suppliers that products contain zero banned substances. Maintain audit trail.
-
Rapid Response System: Monitor ANSM press releases, DGCCRF enforcement notices, and RASFF alerts for emergency bans (compounds can be prohibited without advance notice).
-
Geographic Blocking: Implement robust systems preventing sale/shipment of questionable products to France, even if legal in origin country.
The Regulatory Cycle Pattern
France has established a predictable prohibition cycle:
- New cannabinoid appears on market (3-6 months popularity growth)
- Consumer adverse event reports to CEIP-A (addiction monitoring centers)
- ANSM committee review (2-4 months)
- Classification as narcotic (effective immediately upon publication)
- New alternative cannabinoid emerges (cycle repeats)
Strategic Implication: Companies relying on “legal alternative” cannabinoids face perpetual regulatory risk. Product portfolios require constant reformulation or pivot to established, clearly legal compounds (CBD, CBG, CBC, CBN under 0.3% THC threshold).
Import/Export: Border Security and Documentation
Customs Enforcement Infrastructure
French customs (Douane) maintains sophisticated detection and interdiction capabilities at all entry points:
Primary Border Controls:
- Northern Border (Belgium/Netherlands): High personnel presence, frequent random searches
- Southern Border (Spain): Focus on large-scale seizures (80% of intercepted cannabis from Morocco via Spain)
- Air/Sea Ports: Advanced scanning equipment, K-9 units, intelligence-driven targeting
- Postal/Courier Services: X-ray scanning, chemical detection, data analytics
Seizure Statistics Context:
- 2019-2024: Cannabis flower seizures increased 340%
- Cannabis seedling seizures up significantly since 2010 (indicating domestic production shift)
- CBD product seizures accelerating due to THC compliance failures
Required Documentation for Legal CBD Imports
Mandatory Shipping Documents:
Certificate of Analysis (COA):
- From ISO 17025 accredited laboratory
- Total THC measurement ≤0.3%
- Full cannabinoid profile
- Heavy metals, pesticides, microbial testing
- French translation certified
- Batch-specific with unique identifier
Phytosanitary Certificates:
- Proof of authorized hemp variety (EU Common Catalogue)
- Certificate of origin
- Organic certification (if applicable)
Commercial Documentation:
- Detailed product description
- Harmonized System (HS) codes
- Declared value (under-declaration triggers investigation)
- Shipping manifests with complete chain of custody
Compliance Declarations:
- Sworn statement: THC content ≤0.3%
- Confirmation: authorized Cannabis sativa L. variety
- Novel Food status disclosure (if applicable)
- Banned cannabinoid negative declaration
Red Flags Triggering Enhanced Inspection
Immediate Customs Escalation:
- Shipment origin from known “gray market” production regions
- Declared value significantly below market average (under-invoicing)
- Incomplete or non-French documentation
- Generic product descriptions (“hemp extract,” “wellness product”)
- Shipper/receiver with prior violation history
- Packaging inconsistent with declared contents
- Multiple small shipments to same receiver (structuring)
Penalties for Non-Compliant Imports
Administrative Penalties:
- Product seizure and destruction (no compensation)
- Storage fees during investigation period
- Customs administrative fines (varies by violation severity)
- Import privilege suspension or revocation
Criminal Penalties (Serious Violations):
- €75,000 fine for narcotic importation
- Up to 5 years imprisonment
- Asset forfeiture
- Criminal record
Business Impact:
- Loss of shipping costs and product value
- Damage to shipper-receiver relationships
- Inclusion in customs watchlists (all future shipments scrutinized)
- Potential insurance policy cancellations
Best Practices for Import Compliance
Pre-Shipment Compliance Audit:
- Retain French regulatory consultant to review documentation
- Conduct independent THC testing by accredited EU laboratory
- Verify all labeling meets French requirements before production
- Cost: €2,000-5,000 per shipment
- ROI: Prevents €50,000+ losses from seized shipments
Customs Broker Partnership:
- Work with specialized customs brokers experienced in CBD/hemp
- Establish pre-clearance protocols
- Maintain rapid-response communication for customs inquiries
- Consider bonded warehouse arrangements for high-value shipments
Documentation Management System:
- Digital repository for all compliance documents
- Version control and audit trails
- Automated expiration tracking for time-sensitive certificates
- Multi-party access for customs, brokers, laboratories
Insurance Coverage:
- Specialized cargo insurance covering regulatory seizure risk
- Business interruption coverage
- Legal defense cost coverage
- Premiums typically 2-4% of shipment value for compliant operators
Enforcement Landscape: ANSM, DGCCRF, and Penalties
Regulatory Authority Structure
France’s cannabis enforcement operates through multi-agency coordination:
ANSM (Agence Nationale de Sécurité du Médicament et des Produits de Santé):
- Primary authority for classifying substances as narcotics
- Medical cannabis authorization and oversight
- Adverse event monitoring via CEIP-A network
- Emergency ban authority (no parliamentary approval required)
- Powers: Product recalls, manufacturing suspensions, criminal referrals
DGCCRF (Direction Générale de la Concurrence, de la Consommation et de la Répression des Fraudes):
- Food safety and consumer fraud authority
- Novel Food regulation enforcement
- CBD product compliance verification
- Mystery shopping operations
- Powers: Product seizures, market bans, administrative fines, criminal prosecution
French Customs (Douane):
- Border control and import/export enforcement
- Cross-border trafficking interdiction
- HS code classification verification
- Powers: Seizure, administrative penalties, criminal referrals
National Police/Gendarmerie:
- Recreational cannabis possession enforcement
- Trafficking investigations
- Organized crime cannabis operations
- Powers: Arrest, search, seizure, prosecution
Enforcement Mechanisms
Administrative Enforcement:
- Fixed penalty notices (€200 possession fines)
- Product recalls and market withdrawals
- Warning letters and compliance orders
- Import/export license suspensions
- Business registration revocations
Criminal Prosecution:
- Possession: €3,750 fine, 1 year imprisonment (aggravated cases)
- Distribution/trafficking: €75,000 fine, 5 years imprisonment
- Favorable presentation of narcotics (Article L3421-4): €75,000 fine, 5 years imprisonment
- Fraudulent labeling: €300,000 fine, 2 years imprisonment
- Novel Food violations: Varies by severity
Market Surveillance Programs:
- Continuous online monitoring of CBD retailers
- Social media advertising compliance checks
- Influencer partnership disclosures
- Age verification system testing
- Mystery shopping (underage purchase attempts)
Penalty Escalation Framework
First Violation (Typically):
- Warning letter with compliance deadline
- Product testing requirement (operator expense)
- Administrative fine (€1,000-15,000)
- Corrective action plan submission
Second Violation:
- Higher administrative fines (€15,000-50,000)
- Mandatory product recall
- Temporary suspension of operations
- Public disclosure of violation
- Criminal prosecution consideration
Third Violation or Serious Initial Violation:
- Criminal prosecution
- Business closure orders
- Personal liability for company officers
- Asset seizure
- Industry ban (permanent or multi-year)
Recent Enforcement Actions (2024-2025)
While specific company names are often not publicly disclosed, enforcement patterns reveal:
CBD Product Seizures:
- Multiple retail chains forced to remove entire CBD flower inventories (THC compliance failures)
- Online retailers shut down for Novel Food violations
- Approximately 40% of mystery-shopped CBD products failed compliance testing
Synthetic Cannabinoid Operations:
- Paris-area raid network: 15 retail locations shut down (HHCPO products)
- Marseille distributor: €200,000 in product seizures (THCP gummies)
- Lyon online operation: Criminal prosecution for trafficking (H4CBD vape cartridges)
Import Interdictions:
- Spanish border: 500+ kilograms CBD flower seized (THC overages, 2024)
- Netherlands shipments: Multiple seizures lacking French COA documentation
- Poland bulk CBD isolate: Contamination with banned cannabinoids
[GDPR & ISO 27001 Compliance Assessment ToolComprehensive tool for security leaders to evaluate GDPR and ISO 27001 compliance and prioritize remediation efforts
- Batch traceability records
- Import/export documentation
- Customer age verification data
- Medical patient information (April 2026+)
- Prescription records and physician credentials
- Reimbursement claim data
Required Security Measures:
Data Encryption:
- AES-256 encryption at rest
- TLS 1.3 minimum for data in transit
- End-to-end encryption for sensitive communications
- Encrypted backup systems with immutable snapshots
Access Controls:
- Role-based access control (RBAC)
- Multi-factor authentication (MFA) mandatory
- Privileged access management (PAM)
- Audit logging of all data access
- Automatic session timeout policies
GDPR Compliance:
- Data processing impact assessments
- Privacy by design implementation
- Right to erasure procedures
- Data breach notification protocols (72-hour CNIL reporting)
- Data Protection Officer (DPO) appointment
2. Supply Chain Attack Surface
Vulnerability Points:
- Laboratory Information Systems: COA falsification risk
- Customs Broker Networks: Documentation tampering
- Shipping/Logistics Partners: Product substitution or contamination
- Retail POS Systems: Transaction data exposure
- E-commerce Platforms: Payment data breaches, credential stuffing
- Vendor Portals: Third-party compromise
Mitigation Strategies:
Vendor Security Assessments:
- Annual SOC 2 Type II audits for critical vendors
- Penetration testing requirements
- Incident response plan reviews
- Insurance verification ($5M+ cyber liability)
- Continuous monitoring and threat intelligence sharing
Digital Certificates and Blockchain:
- PKI infrastructure for document authentication
- Blockchain-based COA verification systems
- Smart contracts for supply chain milestone validation
- Tamper-evident digital seals
Network Segmentation:
- Separate networks for compliance data, corporate IT, production systems
- Zero-trust architecture implementation
- Micro-segmentation for critical assets
- IDS/IPS deployment at all network boundaries
3. Threat Intelligence and Monitoring
Specific Threat Landscape:
Regulatory Spoofing: Attackers impersonating ANSM, DGCCRF, or customs officials via:
- Phishing emails demanding “immediate compliance actions”
- Fraudulent inspection notices requesting facility access
- Fake regulatory penalties requiring payment
- Counterfeit laboratory testing services
Competitive Intelligence Theft:
- Formulation data exfiltration
- Customer list theft
- Pricing strategy reconnaissance
- Regulatory filing espionage
Ransomware Targeting: Cannabis businesses targeted due to:
- Perceived limited law enforcement support
- High-value trade secrets
- Customer privacy concerns limiting disclosure
- Operational disruption sensitivity (perishable inventory)
Recommended Defenses:
Security Operations Center (SOC):
- 24/7/365 monitoring (in-house or MSSP)
- SIEM platform with cannabis industry threat feeds
- Automated incident response playbooks
- Quarterly tabletop exercises
- Annual red team assessments
Threat Intelligence:
- Subscribe to cannabis industry ISACs
- Monitor dark web for credential leaks
- Track regulatory authority communication channels
- Participate in industry information sharing groups
4. Incident Response Planning
Regulatory Incident Scenarios:
Scenario 1: COA Data Breach
- Exposure of laboratory test results showing historical THC compliance failures
- Required actions: CNIL notification (72 hours), affected customer notification, regulatory self-disclosure
- Legal exposure: Product recall orders, administrative fines, criminal investigation
Scenario 2: Customer Database Compromise
- Medical patient PII exposed (April 2026+ risk)
- Required actions: Immediate CNIL/ANSM notification, patient notification, law enforcement coordination
- Legal exposure: GDPR fines (4% global revenue or €20M), civil litigation, licensing suspension
Scenario 3: Supply Chain Attack
- Ransomware encrypts batch traceability records
- Required actions: Production halt, customs notification (for in-transit shipments), alternative documentation reconstruction
- Business impact: Revenue loss, shipment delays, regulatory scrutiny increase
Incident Response Requirements:
- Documented IR Plan: Updated quarterly, tested annually
- Breach Coach: Retained French privacy law firm on retainer
- Forensics Capability: Digital forensics team (internal or contracted)
- Communication Templates: Pre-drafted notifications for regulatory authorities, customers, media
- Insurance Activation: Cyber insurance policy with regulatory defense coverage
5. Physical Security Integration
Facility Security Standards:
For medical cannabis operations (April 2026+), facilities must meet pharmaceutical-grade security:
Perimeter Security:
- Fenced boundaries with intrusion detection
- Vehicle access control (license plate recognition)
- Security personnel (24/7 for indoor cultivation facilities)
- Lighting standards (minimum 1.0 foot-candle at fence line)
Access Control:
- Biometric authentication (fingerprint or iris scanning)
- Two-person integrity zones for storage areas
- Visitor management systems with government ID verification
- Access logs retained 3+ years
Video Surveillance:
- 24/7 recording of all production, storage, and access areas
- Minimum 1080p resolution
- 90-day retention minimum (180 days recommended)
- Tamper-evident storage
- Law enforcement access protocols
Inventory Management:
- RFID or barcode tracking at unit level
- Daily reconciliation requirements
- Variance investigation protocols (>0.5% triggers audit)
- Destruction documentation with dual verification
Integration with Cybersecurity:
- Surveillance systems on isolated VLANs
- Access control integrated with identity management
- Automated alerts for unusual access patterns
- Physical-digital convergence for incident correlation
Future Outlook: 2026 Medical Expansion and Regulatory Trends
April 2026: The Great Transition
Immediate Changes Expected:
Patient Access Expansion:
- Current pilot: 3,209 patients (as of March 2026)
- 2026 Projections: 5,454 patients (government estimate)
- 2027 Projections: 15,000-25,000 patients (industry consensus)
- 2035 Projections: 457,129 patients, €806M market (Prohibition Partners)
Prescriber Availability:
- March 2026: 2,291 trained healthcare professionals (531 specialists, 213 GPs, remainder hospital staff)
- Critical Gap: 200+ patients per prescriber if 2035 projections realized
- HAS Expansion Plan: Simplified training pathways, online certification modules, pharmacy-integrated education
Reimbursement Decision (Q1 2026):
- Optimistic Scenario: 60-80% reimbursement rate, €806M market by 2035
- Pessimistic Scenario: “Insufficient medical value” designation, <€100M market, patient access collapse
- Most Likely: Tiered reimbursement (80% for epilepsy/MS, 50% for pain, 30% for palliative), €300-500M market by 2035
Regulatory Trends to Monitor
1. Neo-Cannabinoid Ban Expansion
Anticipated classifications in 2026:
- THV+ and THV-N10 (likely Q2-Q3 2026)
- 10-OH-HHC and 8-OH-HHC metabolites
- CBN above certain thresholds (under ANSM review)
- Generic “psychoactive cannabinoid analog” classification similar to U.S. Federal Analogue Act
2. CBD Classification Uncertainty
ANSES Reproductive Toxicity Review:
- Proposal: Classify CBD as “presumed human reproductive toxicant”
- ECHA Public Consultation: Closed May 16, 2025
- Committee for Risk Assessment: Opinion expected Q1 2026
- Potential Impact: Pregnancy warnings, sales restrictions, marketing limitations
- Industry Response: Coordinated scientific rebuttal, alternative safety data submission
3. Novel Food Regulatory Pathway
Timeline Expectations:
- First CBD Novel Food authorization: Unlikely before 2027
- Application backlog: 100+ pending applications (EU-wide)
- EFSA bandwidth: 3-5 authorizations per year maximum
- French Market Impact: Ingestible CBD market remains suppressed 2026-2028
Alternative Compliance Routes:
- Traditional Food Status: Demonstrating pre-1997 consumption history (extremely difficult for CBD)
- Cosmetic Classification: Non-ingestible products with beautification claims
- Pharmaceutical Pathway: Medical cannabis framework for high-CBD products (minimal THC)
4. Advertising and Marketing Restrictions
Expected Tightening:
- Explicit health claim prohibitions expanding to “implied wellness benefits”
- Influencer marketing regulations with mandatory disclosure and content review
- Age verification requirements for all digital cannabis content (18+ or 21+)
- Geographic targeting restrictions (limiting cross-border e-commerce)
- Packaging plainness requirements similar to tobacco products
5. International Harmonization Pressures
France’s regulatory approach increasingly isolated within EU:
Permissive Jurisdictions Creating Arbitrage:
- Netherlands: HHC and neo-cannabinoids still legal (as of September 2025)
- Spain: Decentralized enforcement, cannabis social clubs tolerated
- Portugal: Decriminalization model with harm reduction focus
- Luxembourg: Recreational legalization in progress
EU-Level Coordination Challenges:
- EMCDDA (European Monitoring Centre for Drugs and Drug Addiction) recommendations often ignored
- Schengen agreement complicates border enforcement
- ECJ rulings favoring product free movement vs. national public health claims
- Cannabis Europa advocacy for harmonized medical framework
France’s Response Strategy:
- Bilateral agreements for enhanced border controls
- Push for EU-wide synthetic cannabinoid bans
- ANSM coordination with German BfArM, Austrian AGES
- Maintaining sovereignty over domestic medical cannabis framework
Strategic Recommendations for Businesses
Short-Term (Q1-Q2 2026):
Medical Cannabis Positioning:
- Pharmaceutical companies: Pursue ANSM Temporary Use Authorizations immediately
- Technology providers: Develop GDPR-compliant prescription management systems
- Security firms: Position for facility security assessment contracts
- Consultancies: Offer physician training program development
CBD Compliance:
- Audit all products for banned cannabinoids (use multiple independent labs)
- Shift to non-ingestible product lines until Novel Food clarity
- Implement real-time ANSM/DGCCRF monitoring systems
- Establish French legal counsel relationships for rapid response
Market Exit Considerations:
- Evaluate France market ROI vs. regulatory burden
- Consider geographic reallocation to permissive EU markets
- Maintain relationship networks for potential re-entry post-2027
Medium-Term (2026-2027):
Regulatory Intelligence Investment:
- Hire full-time regulatory affairs specialists (French-fluent)
- Subscribe to professional regulatory tracking services
- Participate in industry associations (UIVEC, CNPC)
- Establish relationships with ANSM, DGCCRF officials (via trade groups)
Technology Infrastructure:
- Implement blockchain-based supply chain verification
- Deploy AI-powered compliance monitoring for advertising/content
- Adopt pharmaceutical-grade ERP systems with audit trails
- Build redundant data storage across EU jurisdictions
Market Diversification:
- Don’t rely solely on French market for EU strategy
- Build parallel operations in Germany, Netherlands, Spain
- Develop country-specific SKUs matching local regulations
- Maintain flexible manufacturing for rapid product reformulation
Long-Term (2027-2030):
Pharmaceutical Integration:
- For medical cannabis: Full integration into pharmaceutical supply chain
- Pursue collaborations with established pharmaceutical companies
- Invest in clinical trial programs for novel cannabinoid therapies
- Build relationships with hospital pharmacy networks
Recreational Legalization Preparedness:
- Monitor political developments (45% public support for legalization as of 2025)
- Develop contingency business plans for potential 2028-2030 legalization
- Establish brand equity and customer relationships despite current restrictions
- Position for rapid scaling if regulatory environment shifts
Sustainability and ESG:
- Indoor cultivation optimization for energy efficiency (regulatory requirement + cost reduction)
- Carbon-neutral supply chain development
- Social equity program development (anticipating potential legalization requirements)
- Transparent sustainability reporting for investor relations
Conclusion: Navigating Complexity with Intelligence
France’s cannabis regulatory environment in 2026 represents one of Europe’s most challenging compliance landscapes. The simultaneous enforcement of strict recreational prohibitions, aggressive synthetic cannabinoid bans, complex CBD regulations, and transformative medical cannabis framework creates a multi-dimensional risk environment requiring sophisticated security and compliance infrastructure.
Key Takeaways:
- Regulation is Dynamic: ANSM can ban substances without warning. Continuous monitoring is not optional—it’s existential.
- Documentation is Defense: Comprehensive, French-compliant documentation is the only reliable protection against enforcement action. Invest in compliance infrastructure before market entry.
- Security is Foundational: The intersection of regulatory data requirements, patient privacy obligations, and supply chain integrity demands enterprise cybersecurity architecture. Budget accordingly.
- April 2026 is Pivotal: The medical cannabis framework transformation represents the largest opportunity for compliant operators and the greatest risk for unprepared businesses.
- Novel Food Paralysis Continues: Ingestible CBD products remain high-risk until EU regulatory pathway clarifies (2027+ timeline).
- Pharmaceutical Standards Apply: Whether medical cannabis or CBD products, France requires pharmaceutical-grade quality systems, security protocols, and documentation standards.
The CannSecure Approach:
At CannSecure.tech, we provide specialized security and compliance services for cannabis businesses navigating complex regulatory environments:
- Regulatory Intelligence Platform: Real-time monitoring of ANSM, DGCCRF, and RASFF enforcement actions
- Supply Chain Security Audits: Third-party assessment of digital and physical security controls
- Incident Response Services: 24/7 support for regulatory and cybersecurity incidents
- Compliance Documentation Systems: Turnkey solutions for COA management, batch tracking, and audit trails
- Training Programs: Cannabis-specific cybersecurity and regulatory compliance education
The French market offers significant opportunity for sophisticated operators willing to invest in comprehensive compliance and security infrastructure. Success requires treating regulatory compliance not as a burden to minimize, but as a competitive advantage to maximize.
About CannSecure
CannSecure.tech specializes in cybersecurity, compliance, and risk management services for the global cannabis industry. Our team combines deep regulatory expertise, enterprise security architecture, and cannabis market intelligence to help businesses navigate the world’s most complex regulatory environments.
Disclaimer: This article provides general information about French cannabis regulations as of January 2026. It does not constitute legal advice. Consult with qualified French legal and regulatory counsel before making business decisions. Regulatory environments change rapidly; verify current requirements before relying on this information.
Sources:
- Agence Nationale de Sécurité du Médicament et des Produits de Santé (ANSM)
- Direction Générale de la Concurrence, de la Consommation et de la Répression des Fraudes (DGCCRF)
- European Commission Technical Regulation Information System (TRIS)
- Haute Autorité de Santé (HAS)
- French Public Health Code (Code de la santé publique)
- Prohibition Partners Market Analysis 2025
- Augur Associates State of Medical Cannabis Industry in France Report
- Cannabis Europa Industry Intelligence
- CannabisRegulations.ai Compliance Platform
Last Updated: January 4, 2026 Word Count: 9,847 words